Method for enhancement of protein expression in Escherichia coli
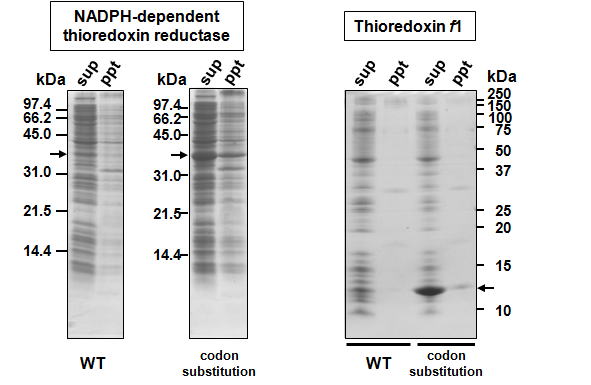
| In vitro studies of protein function generally require expression of recombinant proteins in Escherichia coli, followed by purification and abundant protein yields. The T7 promoter system and other methods have been developed for overexpression of recombinant proteins, although the yields are often lower than desired. In order to improve the efficiency of recombinant protein in E. coli, we introduced A/T-rich sequences downstream of the translation initiation codon, since high G/C content at 5' end of the transcribed mRNA reduce or stop protein translation by these secondary structures. In several cases, we were able to overexpress a recombinant protein by introducing A/T-rich sequences downstream of the translation initiation codon. The region downstream of the translation initiation codons was substituted with an A/T-rich sequence that retained the original amino acid sequence in target proteins. By this method, we expressed recombinant proteins at high levels in E. coli. |
|||
 |
|||
 |
|||
More information is provided in the following reference. Please cite this paper, if you use the codon substitution method for enhancement of recombinant protein expression. Ken MOTOHASHI and Yuki OKEGAWA "Method for enhancement of plant redox-related protein expression and its application for in vitro reduction of chloroplastic thioredoxins" Protein Expr. Purif. 101, 152-156 (2014) Notes ※You should introduce A/T-rich silent mutations downstream of the translation initiation codon, corresponding to 7–10 amino acid residues from N-terminus. ※You should avoid using minor codons in E. coli. ※In several cases, we failed to improve the efficiency of recombinant protein expression by using the codon substitution method to introduce A/T-rich sequences downstream of the translation initiation codon, although the mechanism of this failure remains unclear. |
|||
 |
||||
| Return to "Technical tips" | ||||
| E-mail: |